Hello Sour Beer Friends!
Today I would like to discuss an important family of compounds responsible for many of the appealing aromas and flavors found within sour beers: Esters. Recently, Cale and I were brewing a round of golden and red sours to serve as blending bases for the beers we intend to take to this year’s National Homebrewer’s Convention. In doing so, we opened up an Erlenmeyer flask of our house Brettanomyces culture to find that it was bursting with bright aromas of pineapple and tropical fruit salad. While these aromas have developed a number of times in our aging sour beers, they were especially intense in this particular instance. The experience has led me to think extensively about esters and their formation. Hopefully you will find the topic to be as interesting as I do!
A Quick Primer on Reaction Concepts
To get started, we need to take a step back and talk about a chemistry concept called reaction kinetics that can be a bit dry, but I will do my best to keep it brief and easy to follow. When we think about the chemical reactions that occur within sour beer, most of what we are thinking about is organic chemistry, the same chemistry who’s rules and concepts govern over life at the molecular level. These are the reactions that define life as we know it, and, in our case, apply to what goes on both inside and in the environment around the yeast and bacterial cells living within our beer. Many of these reactions involve the transformation of one type of molecule into another. Living cells perform these transformations for a variety of reasons. These include the release or storage of energy, the production of components that will allow a cell to grow, multiply, or perform special functions, as well as the detoxification or removal of waste products created by the cell.
Any chemical reaction requires some energy from the surrounding environment in order to get started. If this energy requirement is sufficiently low, a reaction will essentially occur spontaneously whenever the starting component or components are present. On the other hand, many reactions require more starting energy than is available in the immediate environment. This situation is typical of many of the metabolic reactions performed by living cells. To accomplish these reactions, cells produce enzymes which lower the energy requirements of their target reaction. In doing so, enzymes allow these reactions to proceed, while the enzymes themselves are not actually consumed by the process. Such reactions can also occur without the presence of an enzyme, but they do so very infrequently.
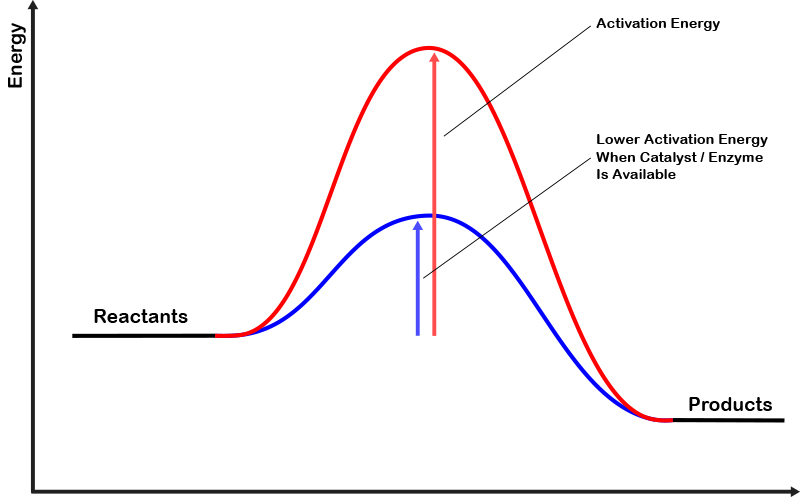
This reaction diagram illustrates how enzymes lower the energy requirements of their target reaction.
The last concept that is important to this primer is the idea that many chemical reactions are two-way or reversible reactions. This means that the products of a reaction can undergo the same process backwards to recreate the starting compounds (reactants). In these cases, what generally determines the ratio of products to reactants in any given environment is the stability of each compound. This stability is directly related to the energy of initiation required for the reaction to proceed in one direction or another. If a reaction has a lower energy of initiation to proceed forward than it does to proceed backwards, then given enough time, the environment will eventually contain an overwhelming majority of products and a very small portion of reactants. It’s important to note that an enzyme that helps a reaction move forward, will not always assist it in moving backwards. A second enzyme may exist which allows a reaction to reverse, and it is through the genetic expression and subsequent production of different enzymes that living cells are able to control what reactions are occurring within them at any given time.
What does all this mean? The important take-away here is that many chemicals in biological situations exist in a state of flux, and changes in either the environment or the enzymes present can influence what compounds exist (and in what ratios) at any given time. Now that we’ve got that out of the way, let’s talk more about esters.
An Overview of Esters
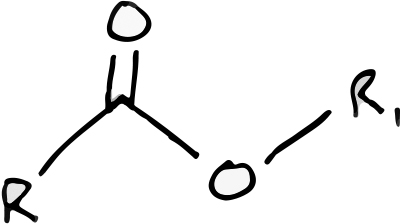
The ester functional group. In this representation R & R1 are any two different carbon-based chains.
Esters, which are technically defined as any chemical containing a specific functional group, are a class of compounds found in a number of biologic scenarios. Fatty acid esters make up a major class of lipids and phosphate containing esters create the backbone of DNA. To give you an idea of the importance of esters in living systems, these are two of the four major classes of biologic macromolecules (the other two being proteins and carbohydrates). The esters that we are concerned with in the aroma and flavor profiles of sour beer are known as low molecular weight esters. These are smaller molecules created from the combination of an acid with an alcohol. Thinking about this last statement for a moment will shed some light on the reason why esters are so prevalent in sour beer, which contains a high proportion of both alcohol and acid!
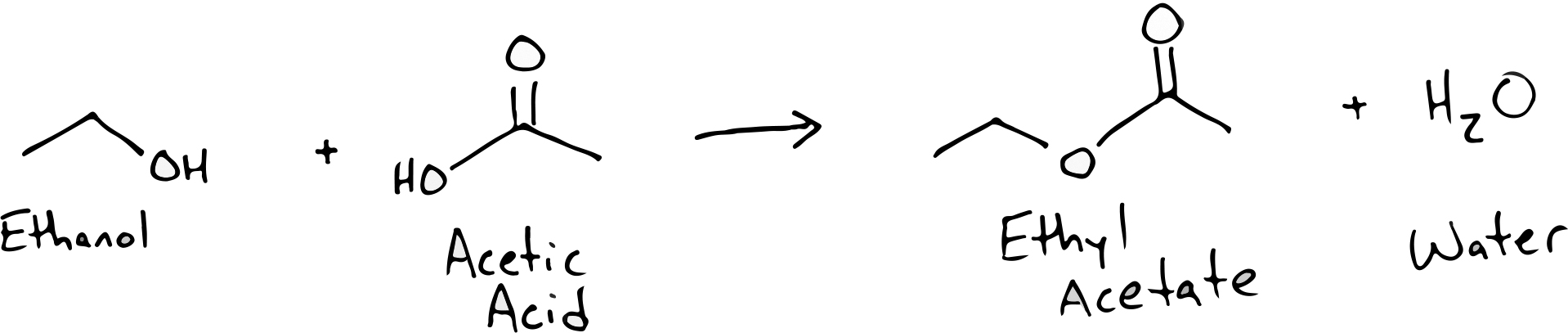
Let’s take a look at an example of how the most common alcohol in sour beer (ethanol) can combine with a common acid (acetic acid aka. vinegar) to produce an off-flavor ester called ethyl acetate:
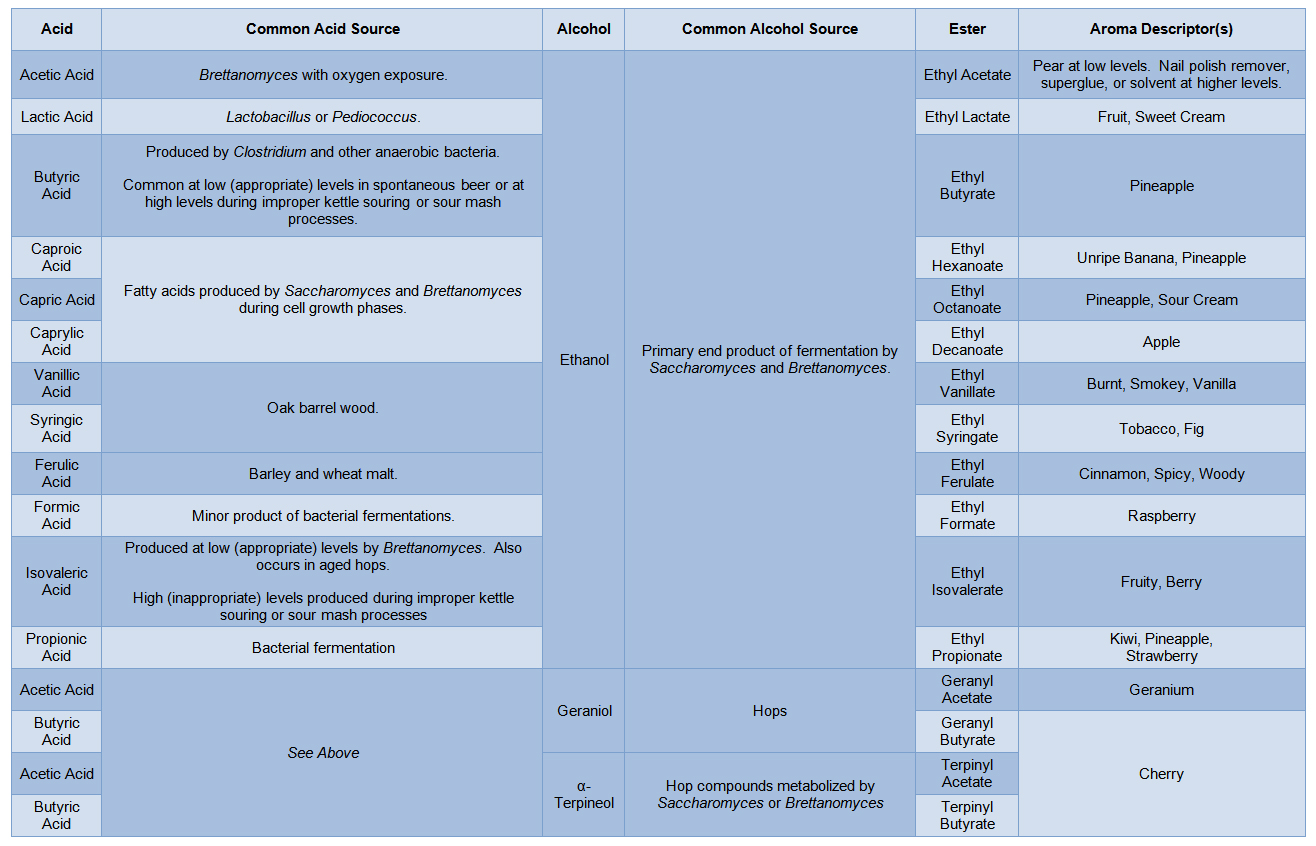
While a sufficient concentration of ethyl acetate can make sour beer smell like nail polish remover, the vast majority of other low molecular weight esters have pleasant aromas and flavors which range from those of flowers, to fruits, to more exotic ingredients such as cinnamon, figs, or tobacco. One of the cool things about these esters is that, in most cases, they don’t just smell “kind of” like their respective fruit or flowers… They smell exactly like them. This is because the same combinations of esters are often being produced by the flowers and fruits themselves to create their signature aromas. It is this fact that has led the perfume industry to use many of these same esters in their fragrances. Before we dive deeper into their production, here is a list of common esters relevant to sour beer:
You’ll notice in the previous table that each ester has an associated acid and alcohol from which it is derived. Therefore, when we are thinking about where a specific ester comes from, we really are asking a three part question:
- Where does the acid come from?
- Which alcohol is it combining with (and if that alcohol is not ethanol, where did that come from)?
And:
- Do these components combine spontaneously or is there an enzyme or catalyst present which helps to drive the formation of this specific ester?
Basics of Yeast Metabolism and Ester Production in Beer
The production of esters is not a process unique to sour beer. In fact, all strains of Saccharomyces and Brettanomyces do produce esters in both varying amounts and varieties during their growth and fermentation processes. Unlike the bacterial sour beer microbes Lactobacillus and Pediococcus, yeast are eukaryotic cells, the same major division of living organisms that all Fungi, Plants, Animals, and Protists fall into. Being members of the eukaryotic club grants yeast not only a full set of intracellular organelles, but also a full range of metabolic pathways including glycolysis, the Krebs Cycle, and mitochondrial (aerobic) respiration. I bring this up because one of the major pathways for the production of esters arises due to the intracellular production of a molecule called Acetyl-CoA. I’m going to go through a brief discussion of yeast metabolism in order to explain why Acetyl-CoA is created within a yeast cell, what its function is, and why its creation can lead to the formation of esters.
When a yeast cell needs energy, one of the best places to obtain it is from the atomic bonds within a glucose molecule. If other sugars are present, these will generally be converted enzymatically to glucose before entering into the first step of a yeast’s metabolic pathway. This first step, called glycolysis, will split a glucose molecule in half, resulting both in the creation of 2 molecules of pyruvate and in the release of energy from the broken atomic bond. At this point, there are two potential pathways that can make use of this pyruvate.
The fermentation pathway, which does not require oxygen, takes these pyruvate molecules and converts them into acetaldehyde and carbon dioxide. A second step converts acetaldehyde to ethanol and leads to the regeneration of vital electron accepting molecules which in-turn allow further rounds of glycolysis to continue. It is interesting to note that, in Saccharomyces, the fermentation pathway is always active if enough glucose is present, even when plenty of oxygen is also available. It is theorized that Saccharomyces ferments ethanol in the presence of oxygen as a way to out-compete other microbes, because ethanol is toxic to many other competitive microorganisms.
Most brewers know that oxygen is vital for healthy yeast cell growth, but many may not fully understand why this is the case. It turns out that yeast cells do not specifically require oxygen to multiply, but they do require it to create the major building blocks of new cell membranes. Much of the pyruvate produced by glycolysis stays in the cytoplasm (liquid inside the cell but between the organelles) and is converted into alcohol as we just mentioned. The remaining pyruvate will undergo a series of reactions which convert it into carbon dioxide and acetyl-CoA. This acetyl-CoA now has one of three fates, two of which require oxygen in order to occur:
- It can enter the Krebs cycle to be further broken down, providing electron carriers with energy to be utilized by the electron transport chain in a process called aerobic respiration. By the way, this process is how we as animals produce the vast majority of our energy and why we need a continuous supply of oxygen to survive.
- It can undergo a process called fatty acid synthesis, which makes use of many 2-carbon Acetyl-CoA units to build long multi-carbon chains. These chains become the fatty acids which connect together to form lipids, a required building block of cellular membranes.
Without oxygen, fatty acid synthesis cannot occur. In these cases, yeast can and will still attempt to multiply and divide, but they have fewer “building blocks” from which to construct themselves. Their cells will be smaller and their cell membranes will be weaker. These weaker cell membranes are more porous, and will leak a variety of chemicals (that are meant to stay within the cell) into the surrounding environment (the beer). These weakened and “leaky” cells result in a variety of off-flavors and give beer a characteristic taste indicative of an “unhealthy” fermentation.
Those of you who are savvy on beer off-flavor compounds will notice that I mentioned a big one in our overview of ethanol fermentation: Acetaldehyde (green apple aroma and flavor). So if acetaldehyde is produced inside the cell, why do we taste it in some beers? The answer here is that the cell membrane is never a perfect barrier and a variety of chemicals that mark “intermediate steps” in a metabolic pathway can leak out of yeast cells and into the surrounding beer. This occurs even when yeast cells are healthy and gives rise to the staggering variety of minor chemicals which define the flavor of beer after fermentation. In the case of acetaldehyde and many other off-flavor compounds, healthy yeast will eventually reabsorb most of these molecules and finish the job converting them into ethanol or whatever they were destined to become. This is the reason why both yeast health and proper resting times on the yeast cake are vital components of a healthy fermentation.
Here is where all of these ideas come together… Just like acetaldehyde, there will be times during fermentation when an overabundance of Acetyl-CoA exists and can also leak out of the cell. This poses a problem for the yeast cell because Coenzyme-A (CoA) molecules are a vital and limited resource that the cell needs to transport not only acetyl groups, but also a variety of intermediate fatty acids during metabolism. Therefore, a yeast cell cannot afford to have excess Coenzyme-A floating around outside of the cell. This is where a special membrane-bound enzyme called Alcohol Acetyltransferase (AAT) comes into play. AAT reclaims coenzyme-A units for the cell by catalyzing the linkage of acetyl groups to ethanol to produce… you guessed it: Ethyl Acetate. This marks the third potential fate of an Acetyl-CoA molecule and this final option is available whether or not oxygen is present.
The enzyme AAT and several related enzymes, simply called Esterases, are the reason why esters are so readily produced by both Saccharomyces and Brettanomyces yeast cells. Furthermore, while AAT is fairly specific to creating acetate esters, different species and strains of yeast can all have slightly different esterases. Each variation of this enzyme seems to have a different level of preference for the types of esters that it will most readily produce. Let’s explore this concept by looking at a common ester called Isoamyl Acetate:
One of the signature characteristics found in beers fermented with Hefeweizen yeast strains is a pleasant banana-like flavor and aroma. This arises from an ester produced in high concentration by these yeasts (and therefore favored by their esterases) called isoamyl acetate. Interestingly, if a beer with a detectable level of isoamyl acetate is then inoculated with and aged upon Brettanomyces, this ester will rapidly disappear, while other esters such as ethyl caproate will rise in concentration. This occurs because the esterification reaction is reversible and the esterases present on the surface of Brettanomyces cells will preferentially break down isoamyl acetate rather than form it.
Conditions That Affect Ester Production
While the strains of Saccharomyces or Brettanomyces present will be the greatest factor determining ester production, there are a variety of conditions and situations that will also increase or decrease ester production:
- Temperature – All enzymes function based upon their physical structure, and this structure is affected by the temperature an enzyme is subjected to. Brewers will be very familiar with this concept in regard to mashing, which uses specific temperatures to optimize the activity of amylase enzymes within the malt. It seems that most esterases reach a peak activity around 77 F. Additionally, higher fermentation temperatures tend to result in more rapid yeast growth, which creates a situation in which a lot of excess acetyl-CoA is being produced, resulting in more ester production. Finally, higher temperatures can affect the varieties of esters being produced by encouraging the production of fusel or “higher” (higher molecular weight) alcohols. Keep in mind that while the esters created from these alcohols may be appealing, the alcohols themselves are considered off-flavors, so any process that churns out more fusel alcohols is also a gamble.
- Oxygenation – As yeast go through their growth phase during a fermentation, the production of acetyl-CoA remains high. If enough oxygen is present, most of this acetyl-CoA will be used for fatty acid metabolism. But if oxygenation is absent or incomplete, a great deal of excess acetyl-CoA will remain, driving ester production.
- Pitching Rate – Again, this all comes down the production of acetyl-CoA. If a wort is under-pitched, yeast can sense this and undergo rapid growth and multiplication which results in a high degree of acetyl-CoA production, driving esters. Conversely, over-pitching will result in negligible cell growth and very little ester production.
- Wort Composition – It turns out that the ratio of nitrogen (amino acids and proteins) to carbon (sugars and carbohydrates) in a wort has a direct stimulating effect on yeast growth and the production of acetyl-CoA. All-malt worts will have a higher ration of Nitrogen to Carbon and this encourages cells to keep making acetyl-CoA even when oxygen levels have dropped, thus resulting in more ester production. Conversely, worts with a high percentage of adjuncts experience reduced ester production.
- Unsaturated vs. Saturated Fats – Unsaturated fatty acids actually have an inhibiting effect on AAT and other esterase enzymes. Therefore, special ingredients in a wort which contain unsaturated fatty acids will reduce ester production. These include most types of nuts including peanuts. Additionally, anything containing vegetable oil will reduce ester production.
- Isovaleric Acid – This off-flavor acid, which can be common in uncontrolled sour mash beers, is a direct inhibitor of AAT. Even though isovaleric acid can technically be esterified, it is uncommon for beers with isovaleric acid off-flavors to improve or transform for the better because this acid inhibits its own conversion into a flavor-positive ester.
Transesterification
Much of what we have discussed thus far has been a groundwork to understand where simple acetate esters come from in all types of beer. However, what I find to be fascinating about sour beers is their ability to develop new and impactful ester profiles months or even years into their aging process. If most esters develop during the initial fermentation process, where do these new esters come from? The answer comes from the concept of transesterification.
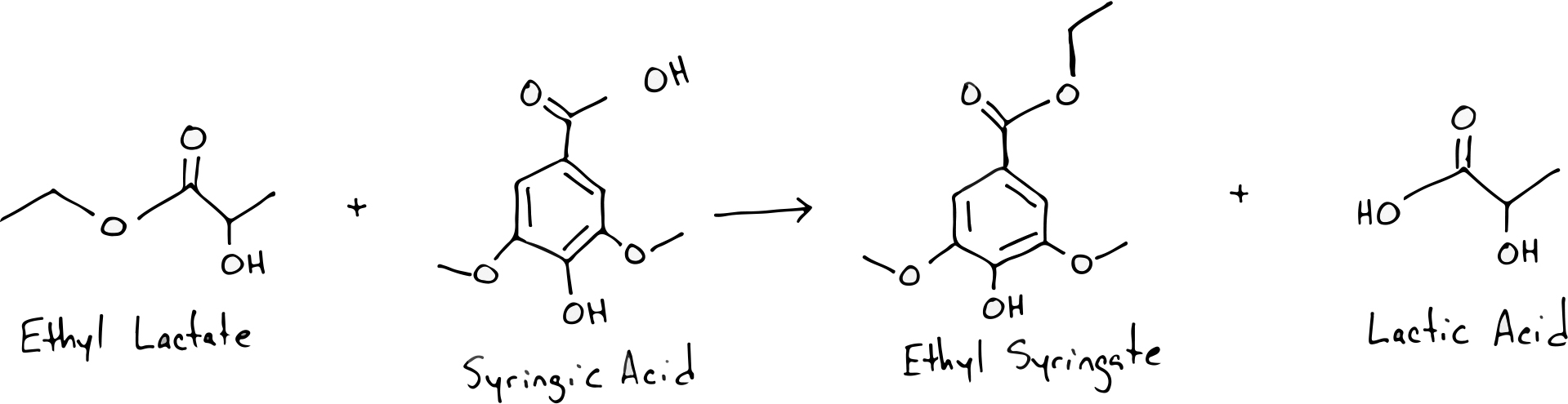
Transesterification occurs when one acid reacts with an ester and in doing so replaces the original acid within that ester. It is through this process that new esters can arise as a sour beer ages, replacing the more common esters such as ethyl acetate and ethyl lactate. Here is an example of how syringic acid (a common wood acid that can enter a beer during barrel aging) can undergo transesterification with ethyl lactate to produce ethyl syringate and lactic acid. Ethyl syringate is a source of tobacco and fig flavors in oak aged sour beers.
This example points out an important idea relating to the complex ester profiles that can develop in sour beers: Many of the components of uncommon and highly appealing esters come from bacterial sources or beer ingredients rather than directly from the yeast fermentations themselves. Here are a few specific examples of esters that arise in this fashion:
- Ferulic Acid is an organic acid present in both barley and wheat malt. Most brewers are familiar with this acid as a precursor for the clove flavored phenol 4-vinyl-guaiacol. However, this acid can also be esterified to form Ethyl Ferulate, which can give sour beer a cinnamon, spicy, and woody character.
- Propionic acid can be produced by a number of bacteria in mixed culture or spontaneous fermentations. While this acid can smell like swiss cheese and body odor by itself, it can become esterified to form Ethyl Propionate which smells like pineapples, kiwis, and strawberries.
- Capric, Caprylic, and Caproic acids are all short chain fatty acids that can be released from yeast and bacterial cells both during growth and upon autolysis. These fatty acids have a mildly rancid aroma reminiscent of goat’s milk (the three make up about 15% of the fatty acids contained within goat’s milk). On the other hand, their resulting esters (ethyl octanoate, ethyl decanoate, and ethyl hexanoate) have a variety of pineapple and tropical fruit aromas. These are the most likely combination of esters that Cale and I were smelling in our Brettanomyces culture, which had been stored at room temperature for several months and was beginning to experience some yeast autolysis.
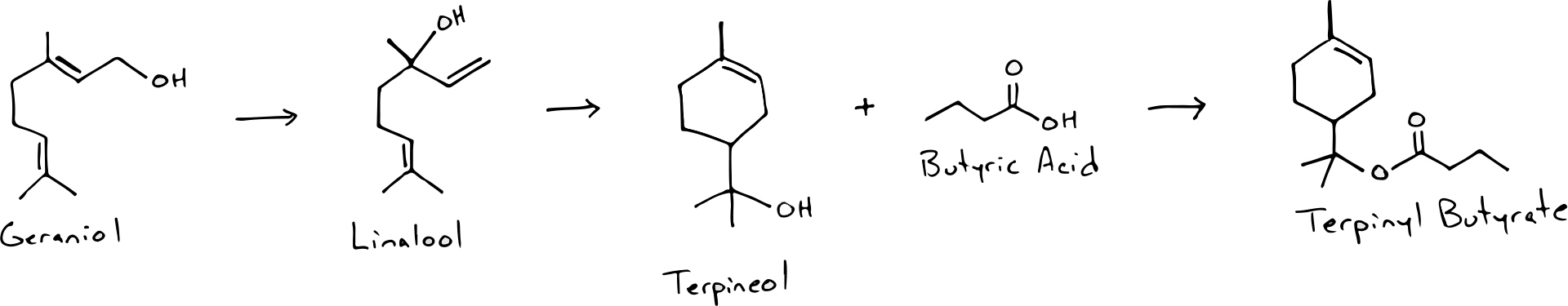
As a final example, I want to walk through a series of transformations that can produce the esters responsible for “cherry pie” aromas in sour beers (that do not contain cherries). Interestingly enough, these compounds actually arise from hops. Many hops contain an aromatic alcohol called Geraniol that smells like roses. During fermentation, yeast can transform this alcohol into Linalool (lavender aroma) and then further into a compound called alpha-Terpineol, which smells like Lilacs. From this point alpha-Terpineol can become esterified via combination with acetic or butyric acid to form Terpinyl Acetate or Terpinyl Butyrate which have a combination of woody, citrus, floral, and fruity characteristics that give the classic “cherry pie” impression.
Conclusion
As brewers, when we think about ester content in our sour beers, it’s important to keep everything in perspective. The goal is not to create the most ester rich beers possible, but rather, when desired, to encourage our strains of Saccharomyces and Brettanomyces to produce a pleasant and potentially unique blend of esters. By understanding where different types of acid and alcohol precursors come from, and how they are transformed into esters, we can make decisions about our ingredients and processes that will encourage the esters we want to form. For example, you will be very unlikely to develop cherry pie esters in a beer made without hops. If you are looking for tropical fruit flavors, strain selection matters, but allowing some Saccharomyces to autolyze can help to free up fatty acids within the beer, enhancing the production of these esters. Understanding that the enzymes that rapidly catalyze ester production are bound to the membranes of yeast cells lets a brewer know that beers are very unlikely to develop further esters after filtration. Thinking along these lines will allow the savvy brewer make decisions that will help steer their sour beers toward their intended goals.
I hope that through reading this article, you will have developed a better understanding of both how and why esters form in all beer, and the factors within sour beer that can drive the creation of unique and impactful ester profiles. Thank you for reading, and as always, please feel free to email or comment here with questions!
Cheers!
Matt “Dr. Lambic” Miller
If you enjoy reading about the science behind various components of sour beer flavor, check out these articles as well!
References
Aquilla, Tracy. “The Biochemistry of Yeast.” MoreBeer. 25 July 2013. Web. 19 Apr. 2016.
Levaj, et al. “Polyphenols and Volatiles in Sour Cherries, Berries and Jams” Food Technol. Biotechnol. 48 (2010) 538–547. Web.
“Brettanomyces.” Milk The Funk Wiki. Web. 19 Apr. 2016.
“Esters in Beer.” Professor Beer. Web. 19 Apr. 2016.
“Estery Flavor.” Malting and Brewing. Web. 19 Apr. 2016.
“FROM ALCHEMY TO SCIENCE: ESTERS, ALDEHYDES, MASS SPECTROMETERS AND HYPER-ACCELERATED AGING.” Cocktail Wonk. Web. 19 Apr. 2016.
Fujiwara, Daisuke, Hiroyuki Yoshimoto, Hidetaka Sone, Satoshi Harashima, and Yukio Tamai. “Transcriptional Co-regulation OfSaccharomyces Cerevisiae Alcohol Acetyltransferase Gene,ATF1 and Δ-9 Fatty Acid Desaturase Gene,OLE1 by Unsaturated Fatty Acids.” Yeast 14.8 (1998): 711-21. Web.
Mauricio, Juan C., Juan J. Moreno, Eva M. Valero, Luis. Zea, Manuel. Medina, and Jose M. Ortega. “Ester Formation and Specific Activities of in Vitro Alcohol Acetyltransferase and Esterase by Saccharomyces Cerevisiae during Grape Must Fermentation.” J. Agric. Food Chem. Journal of Agricultural and Food Chemistry 41.11 (1993): 2086-091. Web.
Peddie, Hilary A. B. “Ester Formation In Brewery Fermentations.” Journal of the Institute of Brewing 96.5 (1990): 327-31. Web.
Takoi, Kiyoshi, Yutaka Itoga, Koichiro Koie, Takayuki Kosugi, Masayuki Shimase, Yuta Katayama, Yasuyuki Nakayama, and Junji Watari. “The Contribution of Geraniol Metabolism to the Citrus Flavour of Beer: Synergy of Geraniol and β-Citronellol Under Coexistence with Excess Linalool.” Journal of the Institute of Brewing 116.3 (2010): 251-60. Web.
“The Fallacy of Instant Bourbon Part II – The Science.” Red White & Bourbon., 03 July 2015. Web. 19 Apr. 2016.
Yoshioka, Kazuo, and Naoki Hashimoto. “Cellular Fatty Acid and Ester Formation by Brewers’ Yeast.” Agricultural and Biological Chemistry47.10 (1983): 2287-294. Web.