Hello Sour Brewers,
Earlier this year, I announced that I would like to focus several of the educational articles published during 2016 on Sour Beer Blog to topics related to the creation of complex, long aged, sour beers. While I love the versatility and simplicity of fast (kettle) soured beers, I have always believed that the true artistry, skill, and magic of sour brewing is exemplified within aged and blended sours. In February, I started this series of articles with a detailed discussion of Blending Sour Beers. Like a Quentin Tarantino movie, this article began our journey at the end of the story, assuming that the reader already had a collection of mature sour beers with varying flavor profiles to choose from. Now, I would like to take a step backwards to an earlier point in the process with a discussion of mixed microbe fermentation and concepts related to the aging and maturation of sour beers.
We will start our discussion with a casting call, a meet-and-greet of sorts with a number of the major microscopic players that will ultimately shape the flavor and aroma profiles of our fermenting and aging sour beers. After gaining an understanding of these microbes, we will check out some different possible approaches as to how we can introduce them into our worts, and what generalities we can expect from differing fermentation schemes. Lastly, we will explore variables we can control as well as some of the objective and subjective measurements that we can make to track these mixed fermentations. Ultimately, these measurements and tastings will determine when our sour beers have matured and become ready to move into the blending process. So with this roadmap ahead of us, let’s get started!
Article Contents
A Sour Beer Casting Call
Saccharomyces, Brettanomyces, Lactobacillus, Pediococcus, and Friends…
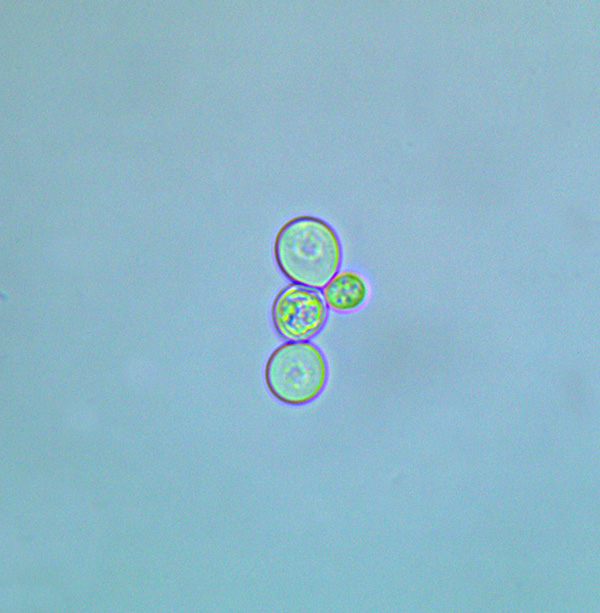
Saccharomyces
Our discussion of microorganisms important to the production of sour beer starts with a family of yeast that all brewers are likely to have some experience with: Saccharomyces (often shortened to “Sacc”). Saccharomyces is a large genus of unicellular yeasts found in a wide variety of environments worldwide. While there are well over a dozen recognized species within the genus, S. cerevisiae (commonly known as “Brewer’s Yeast”) is by far the most widely studied species in the group. In fact, during the explosive growth of biotech science in the 1990’s, S. cerevisiae was the first eukaryotic organism to have its genome fully mapped.
As a general rule, when I think about abstract concepts like microscopic organisms and biochemical reactions, I love to use a lot of metaphors and anthropomorphize things. To this end, I think of Saccharomyces as the dairy cows of the brewing world. Compared to all of the other microbes we will discuss, Saccharomyces cells tend to be physically the largest. Brewing strains of these species have been domesticated since before recorded history to do one specific job and do it very well. That job is to consume a given set of sugars as fast as possible, output a mix of desirable byproducts, and then become sleepy and get out of the way so we can enjoy the benefits of this whole operation in the form of a finished beer, wine, or other fermented beverage.
While multiple species and countless strains of Saccharomyces exist both commercially and in the wild, the key differences between them tend to fall into a few basic categories. The first three of these differentiators we have already mentioned:
- What sugars can the species / strain ferment?
- What products does the species / strain produce? The specific mix of fermentation byproducts created by a given strain is one of the biggest impactors of any beer’s aroma and flavor profile.
- How well does the species / strain flocculate? The length of time that a strain stays in suspension is one of the major determiners of attenuation. If two strains can ferment the same types of sugars, the less flocculant strain will generally be the more attenuative one.
- Lastly, stains will differ in the specific environmental conditions at which they perform their various functions or to what extent they perform them. In more plain terms, this simply means that factors like temperature, pH, and wort composition will affect the performance and behavior of differing strains to differing degrees.
About a decade ago, it was unclear to many brewers whether or not non-Saccharomyces yeasts were even capable of fully fermenting a beer on their own. However, since that time, it has become apparent that Brettanomyces, and other more esoteric yeast families, can not only be used on their own, but can produce delicious and complex beers when used as a sole fermenter. In fact, the popularity of Brettanomyces as a brewing organism has become so widespread that many first time sour brewers opt to use it alone as their yeast choice. That being said, I think that it’s important for sour brewers not to write-off Sacc entirely for its sexier cousin Brett!
Saccharomyces strains can offer several important contributions to sour beer that may be difficult to achieve with Brettanomyces alone:
- When Saccharomyces perform a fermentation, one of the most abundant byproducts (second only to alcohol and carbon dioxide) is a sugar-alcohol called glycerol. Glycerol provides a significant amount of the overall viscosity and body in both beers and wines. It is the fact that Brettanomyces are not capable of producing this compound that can leave 100% Brett fermented beers with an extremely thin and watery mouthfeel. While degree of attenuation is often cited as the source of this characteristic, in reality glycerol is a greater contributor to body than residual sugar (dextrin) content. This is demonstrated by dry red wines, which will often achieve 100% attenuation while retaining a full body due to their high levels of glycerol.
- When Saccharomyces ferments before (or in conjunction with) Brettanomyces, the combination can yield a wider variety of potential flavors than the Brettanomyces may be able to create on their own. This stems from the concept that many of the flavor molecules produced by Sacc will later be transformed into new compounds by Brettanomyces. Without the presence of Saccharomyces, a given strain of Brett may not produce as wide of a variety of flavor and aroma compounds. Therefore, the inclusion of both Sacc and Brett into a fermenting and aging beer tends to increase the overall complexity of that beer.
In single strain “clean” beer fermentations, Saccharomyces health, pitch rate, and temperature stability are paramount factors related to beer quality. In mixed fermentations, Saccharomyces will play only a portion of the role in determining the quality and flavor of the final beer, but it is still important to keep these yeasts as happy as possible at any point during the process when they are actively fermenting.
Brettanomyces
Brettanomyces, or “Brett”, is a family of unicellular yeasts that diverged from a common ancestor of modern Saccharomyces around 200 million years ago. In nature, these yeasts are common inhabitants both upon the skins of fruits and inside fallen and rotting fruit. While many brewers think of Brettanomyces as a “wild” yeast, in reality, many of the strains that have been isolated from breweries and wineries have undergone the same type of accidental domestication process to which Saccharomyces species have been subjected throughout history. This is evidenced by the fact that, unlike strains isolated from Belgian lambic beers or English stock ales, many wild-collected strains of Brettanomyces lack the ability to fully ferment wort. Therefore, for the purposes of this article, we will discuss the characteristics of Brettanomyces strains common to brewing.

A variety of Brettanomyces cell sizes and morphologies can be seen in a 400x magnification of The Yeast Bay’s Brussels Brett Blend.
If Saccharomyces are the dairy cows of the brewing world, Brettanomyces could be thought of as deer. They can be raised for their meat, but they’re more wild and unpredictable than cows. In comparison to Sacc, most Brett strains have physically smaller cells, can consume a wider variety of nutrient sources, and tend to be less predictable in terms of their flavor impact. Brettanomyces cells are not adapted for flocculation like many strains of Saccharomyces. This fact combined with their small cell size allow many strains of Brettanomyces to be highly attenuative. It is not uncommon for mixed culture beers that contain Brettanomyces to become nearly 100% attenuated. Additionally, many Brettanomyces strains have adaptations that allow them to survive long periods of time in very low nutrient environments.
One of the metabolic features of Brettanomyces that is of particular relevance to sour brewers is its ability to convert ethanol to acetic acid in the presence of oxygen. While this ability can become problematic if oxygen exposure is not controlled, it is also likely the reason that Brettanomyces have evolved a tolerance for acidic environments. While many strains of Saccharomyces begin to experience fermentation problems at pH levels below 3.5, Brettanomyces can generally continue fermenting down to a pH of 3.0 or lower.
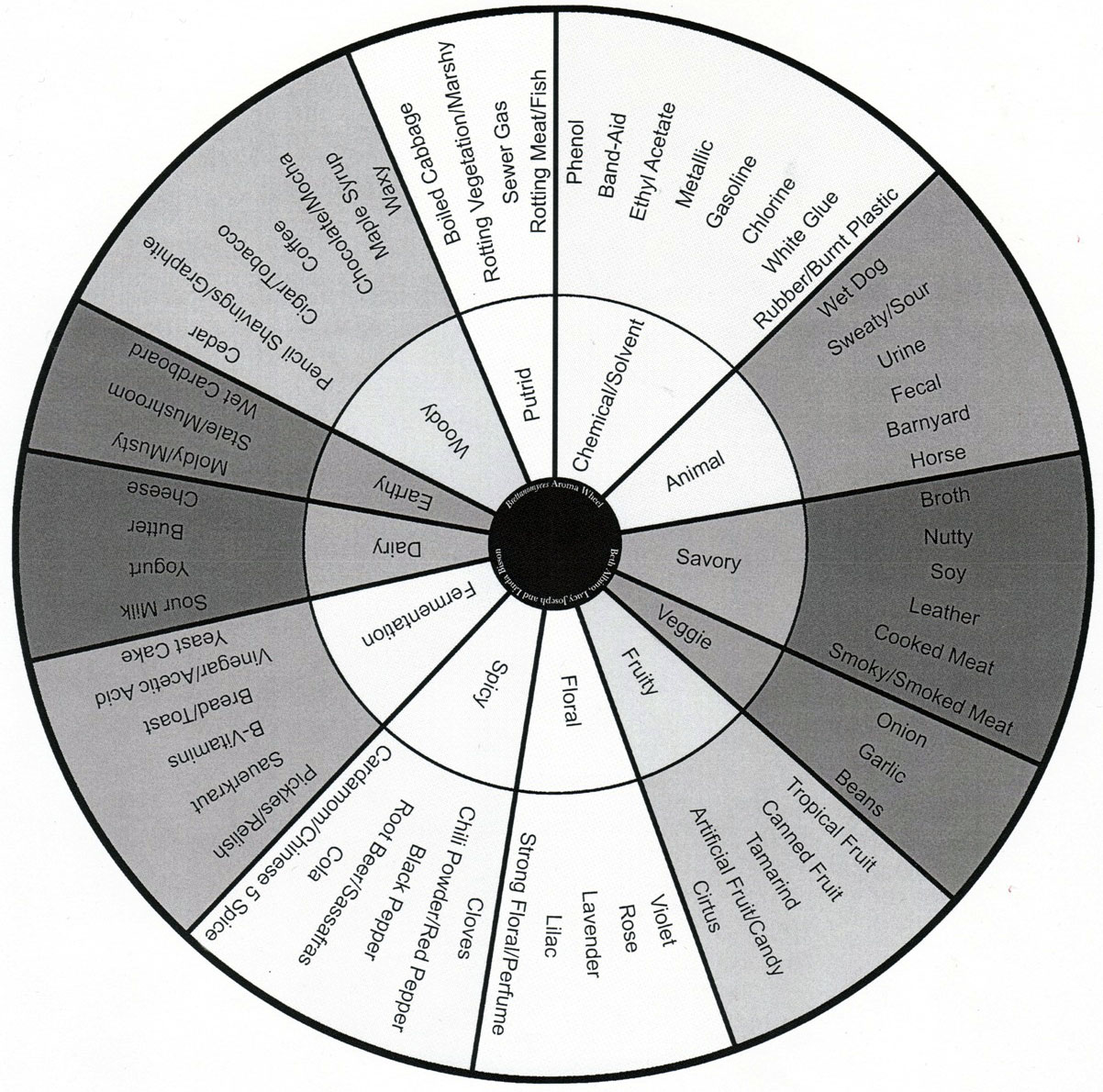
During fermentation, Brettanomyces produce ethanol, carbon dioxide, and acetic acid in a higher concentration than other byproducts. Although many Brett strains make great primary or single strain fermenters, it is their ability to create a wide range of secondary flavor and aroma metabolites that make them indispensable additions to mixed culture fermentations. A number of these unique flavor and aroma descriptors are included in the Brettanomyces Flavor Wheel developed at UC Davis. I have referenced this wheel frequently on the site due to its logical organization and usefulness during beer tastings or blending sessions.
One of the most striking and easily visible characteristics of Brettanomyces is its ability to produce a pellicle on the surface of a beer during fermentation and aging. While not the only genus of yeast or bacteria capable of creating these structures, Brettanomyces tends to be the organism most commonly responsible for their presence under average sour brewing conditions. Pellicles are created at the interface between fluid and air with their hypothesized purpose being to help the cells access oxygen more readily. They are composed of both living and dead cells, as well as a matrix of extracellular protein and carbohydrate structures. Some brewers believe that the formation of a pellicle affects the flavor of the final beer. This may be true in the sense that pellicles tend to form in the presence of some oxygen, but I do not believe that any direct correlation between pellicle formation and beer quality exists. Likewise, I believe that the concept that a pellicle should not be “broken” by sampling or agitation of the beer is a myth. This is also not to say that a process which breaks a pellicle cannot affect beer quality, just that the breaking of a pellicle is irrelevant in comparison to other effects that such actions may have.
Thousands of years of domestication have had their effect on Saccharomyces brewing strains in the sense that they tend not to be survivalists. These strains require proper pitching rates and environmental stability to ensure healthy fermentations and even once these conditions are provided, the vast majority of Saccharomyces cells will begin to die within weeks to months after a fermentation is complete. Brettanomyces, on the other hand, seem to have maintained or potentially been adapted to superior survival ability. Concepts like pitching rate and temperature control have significantly less importance to the performance of Brettanomyces in mixed culture fermentations. While all of these factors do seem to affect the overall characteristics created by Brettanomyces, these yeasts tend not to be prone to problems encountered with improper Sacc fermentations such as under-attenuation, diacetyl and acetaldehyde production, or that “amateur homebrew” flavor that tends to be a hallmark of poor Saccharomyces management.
Brettanomyces vs. Dekkera: What’s the deal?
Some readers will have noticed that the genus name Brettanomyces is sometimes used interchangeably with the name Dekkera. A simple Wikipedia search will indicate that Brettanomyces refers to the anamorph stage of life while Dekkera refers to the teleomorph stage of life. But what does that actually mean?
Here’s the deal: Both Brettanomyces and Saccharomyces are members of a fungal phylum called Ascomycota, and one of the hallmarks of this classification is the fact that these fungi can reproduce not only vegetatively (through budding: this is the common asexual method of yeast reproduction during fermentation) but also sexually through the formation of spores. During favorable conditions like those found early in fermentation, both families of yeast simply reproduce by repeatedly creating clones of themselves. However, when conditions become unfavorable, such as after all of the nutrient sources run out, these yeast can mate through the fusion of two cells and then transform into a structure called an ascus, which will typically contain 4 ascospores. These spores can be thought of as fungal “seeds”: they’re tough and can survive long periods of being dried-out and without nutrients. In fact, the ascus structure typically splits open and releases the spores after the medium the fungi have been living in dries up entirely. This releases the spores into the open air to float to a hopefully new and nutrient rich environment. In the wild, this occurs frequently when a food source such as rotting fruit runs out of sugar and eventually dries up.
It’s important to remember that this mechanism is the natural (undomesticated) state for both Saccharomyces and Brettanomyces, but that the controlled fermentation of beer is a human process. Through domestication, most brewing strains of both Saccharomyces and Brettanomyces have either lost their ability to sporulate entirely or have had these functions significantly diminished. Most strains of Saccharomyces will flocculate and eventually autolyze after fermentation rather than produce spores. In the case of Brettanomyces, although spore formation is a fairly rare event, it is believed that most strains of Brettanomyces (Dekkera) bruxellensis and B/D. anomala retain the ability to create ascospores. On the other hand, no teleomorphic (spore forming) stage has ever been observed for the species Brettanomyces custersianus, B. naardenensis, or B. nanus.
Lactobacillus
Unlike Saccharomyces and Brettanomyces, which are both eukaryotic members of the fungi kingdom of life, Lactobacillus is a genus of rod-shaped prokaryotic bacteria. With well over 100 recognized species, Lactobacillus occur in a number of biologic niches, including several sites upon and within the human body. In addition to their role in sour brewing, these bacteria are integral to the creation of a number of fermented foods such as sauerkraut, yogurt, cheese, kimchi, pickles, kefir, and sourdough bread. These bacteria create lactic acid from the fermentation of simple sugars via one of two metabolic pathways: homofermentative and heterofermentative. During homofermentative metabolism, 6-carbon sugars such as glucose or fructose are split and converted into two molecules of lactic acid. Heterofermentative fermentation allows for these bacteria to utilize sugars with differing numbers of carbons such as arabinose or ribose (5 carbons) and yields lactic acid, carbon dioxide and ethanol or acetic acid. Certain species of Lactobacillus can only utilize one pathway or the other (obligates), while some species can switch between the two depending upon their conditions (facultative).
The sugars that Lactobacilli can ferment vary significantly between species and potentially within strains of any given species. As a general rule though, Lactobacillus are fairly picky fermenters, preferring glucose and occasionally being capable of processing fructose, maltose, sucrose, or other wort sugars. Regardless of their metabolic capabilities, Lactobacillus cannot fully attenuate wort as a single brewing organism. In my farm analogy, I actually think of Lactobacillus like butterflies visiting the crops as they flower. These little guys are delicate in comparison to both yeast and some of the other potential bacteria involved in sour brewing. If conditions are just right, they will flourish, but they don’t compete well with other organisms and certain environments (like any wort with above 5 to 10 IBUs) will knock most strains out entirely.
This is not to suggest that Lactobacillus are not important to sour brewing, only that the conditions in which they will have the most impact are very specific! I personally am a huge proponent of Lactobacillus-first fermentations (kettle souring or quick wort souring) in order to produce a number of fast soured styles such as Berliner Weisse or Gose. I have previously written extensive articles about how to use these processes, check them out here. Fast souring aside, Lactobacillus can also play a vital role in the creation of aged, mixed-culture sour beers as one of the two major genera of lactic acid producing bacteria relevant to brewing.
Due to their popularity as a quick-souring microbe, many people have mistakenly believed that all of the acidity that Lactobacillus can offer to a sour beer must be achieved early on in the process. Fortunately, this is not the case. In my experience, the more accurate concept would be that Lactobacillus must either establish their cell counts early, or have an appropriately large number of cells pitched to begin with, in order to produce significant acidity when in competition with yeast fermenters. For example: I have brewed a number of batches of mixed culture sour beer using the White Labs strain of Lactobacillus delbrueckii. This is a strain known to be notoriously bad at achieving fast acidification for styles like Berliner Weisse or Gose. In my beers, this microbe was pitched into worts with an average pH of 5.2 and held at 110° to 115° F for anywhere from 2 to 4 days. After this time, the pH of the worts would range anywhere from 3.8 to 4.2 on average. In my process, these beers are never re-boiled to kill the Lactobacillus. Rather, they are cooled, oxygenated, and pitched with Saccharomyces to achieve standard attenuation. They are then racked off the yeast cake, and inoculated with a low cell count pitch of Brettanomyces and allowed to age in glass carboys for anywhere between 3 to 12 months on average. Of the several dozen batches that have utilized this fermentation scheme (varying the species and strains of Lacto, Sacc, and Brett), every one of them eventually achieved a final pH of 3.1 to 3.4 (with a robust acidity when tasting). Keep in mind that, in these batches, Lactobacillus was the only acid producing bacteria and the bulk this acidity was achieved after the addition of yeast fermenters.
On the other hand, there have been numerous accounts over the years of brewers who’ve had a fully fermented and attenuated beer, to which they added a (likely low cell count) pitch of Lactobacillus, only to discover that this beer never developed any notable acidity regardless of the aging period. It was actually the number of these reports that led me in 2010 to begin experimenting with Lactobacillus-first mixed culture fermentations. While this fermentation scheme (which has been referred to as my Three Step Method) works wonderfully to produce complex and assertively sour beers with age, I do believe that you could potentially get Lactobacillus to acidify a beer after standard attenuation given that two conditions are met:
- The cell count would have to be high. It’s hard to say how many cells are needed but it must be more than the 2 billion or so packaged in a White Labs vial.
- Brettanomyces must be present in order to liberate fermentable sugars for the Lactobacillus to convert to lactic acid. Brettanomyces excrete the enzymes alpha and beta glucosidase extracellularly (outside of their cells, ie. into the beer). These enzymes are responsible for the breakdown of dextrins (long chain non-fermentable sugars) into glucose. It is the action of these enzymes that allow Brettanomyces to attenuate wort to a significantly higher degree than Saccharomyces brewing strains. Because the chemical breakdown of dextrins is occurring outside of the cells, the liberated glucose can be just as easily consumed by Lactobacillus as by Brettanomyces itself or any other microbes present. There are a few known strains of Lactobacillus that can produce their own extracellular amylases and glucosidases. Such strains would be able to continue to acidify attenuated beer without the presence of Brettanomyces.
Pediococcus
In terms of brewing science, Pediococcus, a family of gram-positive lactic acid bacteria, may be the least studied organisms common to sour beer production. This genus, closely related to Lactobacillus, can be found naturally in a number of environments including upon the surfaces of plants and fruits. Several Pediococcus species are integral to the production of fermented foods such as cheeses and sausages. Some species of Pediococcus have been of interest to both food and medical researchers due to their ability to synthesize a class of antibacterial molecules called pediocins. Pediocins have antibacterial activity against some of the nastier food borne pathogens such as Listeria, Clostridium, and Enterococcus which makes them an important class of natural food preservatives. To wrap up my farming metaphor, I think of Pediococcus like goats. For the most part, these goats produce beneficial products like milk (lactic acid), but in some cases they can break loose, tear up the farm, and crap all over the place (diacetyl, EPS).

“Sour Weapon” from Bootleg Biology is a new strain of P. pentosaceus that can be used to quickly sour wort.
While most Pediococcus species prefer warmer environments and tend not to grow well in beer, the species P. damnosus grows optimally at 72° F and is intolerant of temperatures above 95° F. Unlike the various strains of Lactobacillus which have become prevalent in American sour brewing, Pediococcus damnosus is the organism responsible for the slow acidification of lambic style beers such as gueuze and kriek. All species of Pediococcus utilize homolactic fermentation, which means they break down sugars into lactic acid and cannot create alcohol or carbon dioxide. Depending on the strain involved, Pediococcus can produce both extracellular amylases and glucosidases. Like Brettanomyces, these enzymes allow this bacteria to continue to attenuate wort beyond the limits of Saccharomyces. In most cases, Pediococcus are relatively slow growing organisms that can produce significant lactic acidity in a beer as it ages. As an exception to this trend, at least one strain of Pediococcus pentosaceus has been shown to be capable of fast-acidification of a beer using the same methods by which Lactobacillus can be used to quickly sour wort. While most species of Lactobacillus cannot tolerate more than a few IBUs in a beer, Pediococcus is significantly more hop tolerant. This gives it the capacity to sour a wider variety of classic beer styles including lambic beers, which gain a substantial portion of their flavor profile from aged hops.
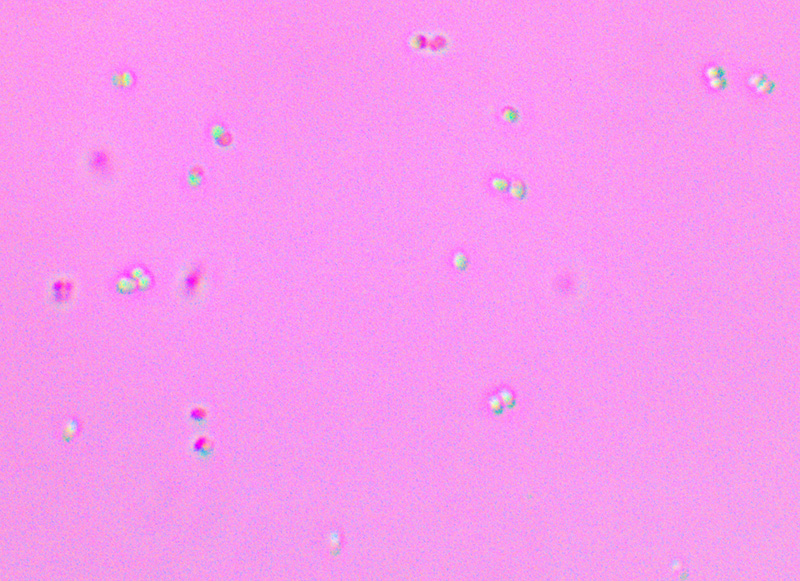
Pushing the limits of my microscope camera, Pediococcus cells can be seen forming clumps of 2 to 4 cells (tetrads) at 1000x magnification.
Pediococcus damnosus is notorious for its propensity to create the off-flavor diacetyl (rancid butter / buttered popcorn / butterscotch) during its acidification process. Therefore, it is always recommended to pair this microbe with Brettanomyces which can clean up this off-flavor as the beer ages. Perhaps the most recognizable, and quite frankly off-putting, characteristic of Pediococcus is its ability to create a viscous extracellular slime within a beer. This substance, often referred to as sickness or ropiness, is a collection of exopolysaccharides (EPS). Exopolysaccharides are long chain carbohydrate molecules that increase the viscosity of the beer, giving it a slick texture and runny appearance (like uncooked egg whites) without producing any significant flavor changes. Luckily, exopolysaccharide existence within a beer is a transient event. Eventually, on a timeframe of weeks to months, enzymes produced by both Pediococcus and Brettanomyces will break this substance down and return a sour beer to its normal viscosity and mouthfeel.
A short video of a beer turned ropy by Pediococcus
Pediococcus has been referenced by a number of sour brewers as creating a more complex flavor profile than Lactobacillus. While the final complexity and blend of flavors in any aged sour beer is dependent on a nearly innumerable number of factors, I tend to believe that Pediococcus beers are not inherently more complex or “better” than Lactobacillus beers. I do agree that fast-sour beers that are ready to serve in a few weeks tend to be significantly less complex than sour beers aged for many months or years. However, given aging time, I believe that Pediococcus and Lactobacillus have similar impacts on the flavor profile of a sour beer with two exceptions:
- Pediococcus can leave a little residual diacetyl which may give Pedio beers a unique flavor profile.
- The fact that Pediococcus is hop tolerant allows for the inclusion of fresh or aged hops in a recipe, increasing complexity in a number of ways.
Less Common Sour Beer Microbes
Enteric Bacteria – Bacteria from genera such as Enterobacter, Citrobacter, Klebsiella, and Hafnia are part of a rod-shaped gram negative family of prokaryotes called the Enterobacteriaceae. While these organisms are not intentionally included in most mixed culture fermentations, their ubiquitous presence in the environment gives them access to beer undergoing traditional spontaneous fermentation. When present, these organisms tend to deplete a beer of free amino acids, leading to sluggish initial fermentation by Saccharomyces. These bacteria metabolize sugars into a variety of potential byproducts including lactic acid, acetic acid, ethanol, and carbon dioxide. Additionally, enteric bacteria influence the flavor profile of a beer through the production of compounds such as DMS (canned corn), indole (fecal), and phenols. These organisms also produce a class of chemicals called biogenic amines. Some of these compounds, when ingested in high quantity (example: an all night sour beer bender!), can have negative health effects such as allergic type reactions and increased blood pressure. Of particular note, and interest to me as a pharmacist, is the fact that patients taking monoamine oxidase inhibitors (MAOIs) are at a particularly high risk of developing hypertensive crisis after the consumption of spontaneously fermented beers, as this enzyme system is used by the body to detoxify biogenic amines.

A “Vinegar Mother” is created by the bacteria Acetobacter aceti and is composed mainly of living cells and cellulose.
Acetobacter – Bacteria from this genus are responsible for the intentional fermentation of vinegar due to their ability to rapidly metabolize ethanol into acetic acid in the presence of oxygen and warm temperatures (above 77° F). They are generally considered an unwanted bacteria in sour beer fermentations for this same reason. While acetic acid plays an important role in the flavor profile of some sour beers, its presence at appropriate levels should be achieved through Brettanomyces activity. If Acetobacter infects a batch of sour beer, it can rapidly produce too much acetic acid. Such beers should be dumped rather than blended, because the inoculation of Acetobacter into the blend can result in the same rapid acetic acid production that plagued the initial beer.
Oenococcus oeni – This ovoid gram positive bacteria, one of only two species in its genus, is commonly used within the wine industry as an agent of malolactic fermentation. It is this ability to convert malic acid to lactic acid which has recently brought this organism to the attention of sour brewers. As a general rule, lactic acid is softer and more palatable than malic acid (the most common acid in many fruits). This process releases carbon dioxide, and will generally result in the transformation of the majority of malic acid into lactic acid in around 2 weeks. Due to the creation of diacetyl from any citric acid present, this process gives wines like chardonnay their buttery flavor, and can impart this same compound to a sour beer, where it is generally considered a flaw. It’s important to note that this organism is not isolated in its ability to perform malolactic fermentation. Many species of both Lactobacillus and Pediococcus also share this capacity.
Oxidative Yeasts – Candida (anamorphic) and Pichia (teleomorphic) yeasts are another group of organisms uncommon to mixed cultures but commonly present in spontaneously fermented beers. These yeasts are pellicle forming and utilize oxygen to metabolize ethanol into acetic acid. Unlike Brettanomyces however, they do not produce alcohol. Rather, these yeast may contribute to the cidery flavors found within lambic beers. When present, these yeast also have a high propensity for the creation of ethyl acetate from acetic acid.
Other Non-Conventional Brewing Yeasts – Recently, both scientists and brewers have shown interest in screening a variety of families of yeast such as Torulaspora, Kluyveromyces, Cyberlindnera, and Wickerhamomyces for potentially unique aroma and flavor profiles. While, so far, none of these genera show the versatility towards beer fermentation that Sacc and Brett have proven, there is still much research to be done.
Bugging Out with Mixed Culture Fermentations
Now that we’ve familiarized ourselves with the most common microbes involved in sour beer fermentations, lets that a look at various ways to manage those fermentations. The goal of this section will be to review an assortment of the most common mixed fermentation schemes and gain an understanding of the control points available for these practices.
Before we dive any further into this topic, I’d like to take a brief sidetrack into the mathematical concept of chaos. Overly simplified, this concept basically states that if any system is sufficiently complex, then very minor alterations in starting conditions can have very large, and difficult to predict, effects on the ending conditions. Because all fermentations involve living organisms, they are inherently complex systems. Sour beer fermentations are notoriously unpredictable because elements like multiple microorganisms, longer aging times, and inconsistency between barrels introduces more variability than exists when brewing “clean” beers. Due to this reality, we can never predict the exact outcome of any single mixed fermentation with any high degree of reliability.
I bring all of this up not to discourage, but rather to emphasize the importance of keeping the big picture in mind when brewing and aging sour beers. Many, if not all, of these beers should be brewed with an expectation that blending will occur to create your final product. Additionally, the unpredictable nature of these fermentations should instill the importance of true “fermentation management” to a sour brewer. Unpredictable does not mean unmanageable. If attention is paid to a sour beer through both its initial fermentation and aging process, several control points will become available to help a brewer course correct a sour beer that is not developing as planned. This attention will also help to identify beers that have developed irreparable off-flavors, so-called “blend killers”, that should be dumped in order to free up both time and resources that would otherwise be wasted upon them.
With these concepts in mind, lets dive into the most common types of mixed culture fermentations available to a sour brewer:
All The Bugs Up Front
Nothing beats this method for sheer simplicity. Basically you add your house mixed culture or whatever combination of cultures you would like to use at the very beginning of fermentation. There are both pro’s and con’s to this approach. The addition of yeast fermenters at the very beginning of fermentation reduces the risk for certain types of off-flavors associated with unwanted bacterial contamination such as isovaleric or butyric acid. On the other hand, these beers tend to develop their acidity more slowly unless a very robust culture of Lactobacillus is included in the mix. In addition to other flavor characteristics, both the timetable for acidification and total ending acidity become greater variables. Most of the lab created mixed cultures have pretty wimpy Lactobacillus or Pediococcus strains included. For this reason, I think this method tends to benefit from the inclusion of some bottle dregs from lambic or other commercial sour beers. The addition of dregs will generally increase overall flavor complexity and speed up souring when using this method. Keep in mind that it may still take 6-24 months for these beers to fully develop.
Control Points:
- Strain Selections – Allows for dramatic changes in fermentation characteristics.
- Pitching Rates – Allows the brewer to increase or decrease the speed of acidification, alcohol fermentation, and potentially the intensity and variety of fermentation characteristics.
- Addition of New Microbes at Any Point During The Beer’s Aging Cycle – Allows the brewer to course correct issues such as diacetyl off-flavor, lackluster acid development, or lack of ester or phenol development.

“Bug Farm” by East Coast Yeast is an example of a complete mixed culture that can be used to produce excellent sour beers.
With the right cultures, this method can make some of the world’s best and most complex sour beers. Because the acidity tends to develop more slowly with this method, Brettanomyces and Saccharomyces tend to produce a wider range and intensity of phenolic compounds, increasing the overall “funkiness” of these beers. This is the most old-school method around for producing sour beer and when a lackluster blend of microbes is employed this method can easily yield lackluster beers with little to no acidity. If this happens, it’s best to pitch a fresh culture of Pediococcus or a nice portion of dregs from beers known to contain Pedio.
Keys to Success:
- Use of a robust and tested house culture or the assembly of a new blend of microbes with at least one yeast fermenter with an adequate cell count to carry out a timely initial fermentation.
- Build variation into your aging stock by altering the microbes used.
- Monitor flavor development while avoiding excess oxygen exposure.
- Have patience. These beers often take 1 to 2 years to fully mature.
Staggered Microbe Additions
In comparison to the method we just discussed, the staggered addition of microbes to a sour beer fermentation is a newer practice that offers a greater degree of control over the final product. The variations within this method all share the concept that if certain microbes are added to a beer at different time points, then the conditions under which they are actively fermenting the beer can be optimized to achieve a variety of results. Variables such as temperature, oxygenation, amount of fermentable sugars, and microbe pitch rate can be individually controlled for each step of the process. In its most extreme form, this method can be used to fast-sour a beer using a robust pitch of Lactobacillus, which is then followed up with Sacc, Brett, or both in order to turn around certain sour beer styles in as little as a few weeks. For more information on fast souring including expanded details on avoiding off-flavors during Lactobacillus fermentations, click here. Despite the popularity of fast souring, the concept of allowing Lactobacillus to ferment in a beer before the addition of yeast fermenters is not a “one trick pony” useful only for brewing low-complexity Berliner Weisses and Goses. This practice can be used to establish a healthy colony of Lactobacillus early in the beer’s fermentation process, even if a high degree of acidity isn’t desired or achieved until later during a beer’s aging. Let’s take a look at a few variations on these practices and review their potential benefits:

Goodbelly (L. plantarum) and a large pitch of Saccharomyces which was used to brew 36 gallons of sour red blending stock.
Lactobacillus –> Saccharomyces –> Brettanomyces
This method, sometimes referred to as my “Three Step Fermentation” is the most common process that I personally use to create sour beers for my blending program. In my opinion, this method can be used to create sour beers with a moderate to high degree of complexity without having to deal with some of the drawbacks of using Pediococcus such as diacetyl production or the possibility of ropiness due to EPS formation. These beers also tend to be ready for blending or consumption on a somewhat accelerated timetable, generally in the 3 to 12 month range.
Control Points:
- Strain Selection – Individual strain choices can be made for each of the microbes to be used. This allows for greater variability in batches (beneficial in blending) and also allows a brewer to identify the impact that changing a single strain in the blend can have.
- Pitch Rate – The results of a mixed culture fermentation can be altered significantly by pitching rate.
- Lactobacillus pitch rates will affect the speed and, potentially, the degree of acidification.
- Saccharomyces pitch rates can be optimized to reduce off flavors such as diacetyl, DMS, or acetaldehyde.
- Brettanomyces pitch rates, in my experience, affect the overall presentation of Brett derived flavors. Low pitch rates into attenuated wort seem to produce more of the classic “lambic” funky barnyard flavors, while higher pitch rates into less attenuated worts seem to result in flavors more commonly found in farmhouse ales and Brett saisons.
- Oxygenation & Oxygen Avoidance – These control points allow for fermentation health and avoidance of unwanted acetic acid production by Brettanomyces. Oxygen levels can be reduced for Lactobacillus fermentation, increased for healthy Saccharomyces growth and fermentation, and kept low for Brettanomyces aging.
- Temperature – The ability to warm a wort will aid the growth of many strains of Lactobacillus, while cooling and maintaining a stable temperature through Saccharomyces fermentation will help to drive attenuation and reduce off-flavors such as acetone and fusel alcohols. Brettanomyces tends to produce a balance of phenols and esters when held between 70 to 80° F. At temperatures cooler than this, Brett characteristics seem to develop more slowly but still maintain a normal balance between esters and phenols. At temperatures above 80° F, many Brettanomyces strains start to behave more like saison yeasts, producing a greater degree of spicy phenols.
- pH – The measurement of pH allows a brewer to control several aspects of the fermentation. The most obvious of these is the sourness of the beer. Less obvious are characteristics such as attenuation, ester, and phenol development. At pH levels below 3.5 the final beer is likely to be assertively sour, but it is also less likely to enjoy a healthy fermentation by Saccharomyces. This can be combated by increasing Sacc pitch rate, or by relying on Brettanomyces to achieve full attenuation and clean up any off-flavors from Saccharomyces. Additionally, beers that are quickly soured to pH levels below 3.5 tend to develop less Brettanomyces funk (phenolic compounds) than beers which develop their acidity later during the aging process.
As a beginning sour brewer, I began experimenting with this three step fermentation process in 2010, and I’ve continued to successfully use it to this day. My scientific side has always been drawn to this method because of the control it gives you over strain choice and pitch rates per strain. I think that this level of control helps to remove some of the larger variables from the sour brewing process and offers more predictability than methods which rely on unknown or un-quantified blends of microorganisms. On the other hand, my artistic side still appreciates the complex beers that this process can feed into my blending program. On a typical sour brew day, I may produce up to 40 gallons of wort to be split into numerous fermentors. Each fermentor will then receive a different assortment of strains for each step in the fermentation. Doing so creates the flavor and aroma variety that I think really helps to make my blending program a success.
Keys to success:
- Monitor aspects of your fermentation such as gravity, pH, and flavor/aroma at each step in the process.
- Keep detailed notes on strain selections, pitch rates, temperatures, lengths of fermentation, and wort/beer measurements. Doing so will help you to identify trends or recreate beers that turned out particularly well.
- For intensely sour “acid beers”, use large pitches of Lactobacillus, select aggressive strains such as Omega Yeast’s L. plantarum or Gigayeast’s L. delbrueckii, and allow these strains to survive throughout the Saccharomyces & Brettanomyces portions of fermentation (no fast-sour second boil). Keep in mind that both of these strains can create worts so acidic, that even Brettanomyces will have difficulty reaching full attenuation.
- For beers with a balance between funky phenols and fruity esters, I like to select slower acidifying strains of Lactobacillus such as White Lab’s L. delbrueckii. These are followed up by fruity Sacc strains such as those used to produce English Ales. I then age these beers on classic strains of B. bruxellensis or anomalus.
- For beers that really maximize the farmyard funk, I again select slower acidifying Lacto strains, but I then follow them up with spicy Saccharomyces strains such as those found in Saisons or Farmhouse Ale Blends. I age these beers with relatively funky Brett strains such as White Lab’s B. bruxellensis “lambicus” or East Coast Yeast’s “Dirty Dozen” Brett Blend.
(The Milk The Funk Wiki maintains an excellent list for selecting your Brettanomyces strains or blends)
Other Staggered Microbe Fermentations:
The staggered addition of microbes can naturally be modified to utilize a greater or fewer variety of microorganisms than the Lacto-Sacc-Brett approach that I’ve just described. Here are a few variations that I feel are worth mentioning:
Lactobacillus –> Brettanomyces (or) Brettanomyces –> Pediococcus
The combination of a single genus of lactic acid bacteria with Brettanomyces is a method which has been used effectively to produce sour beers. If choosing a Lactobacillus first approach, the degree of initial acidification will have effects upon the final presentation of “funk” as described earlier. The Pediococcus last approach has the benefit of allowing Brettanomyces to be more expressive, but also carries the potential for longer maturation times.
Lactobacillus –> Saccharomyces –> Brettanomyces +\- Pediococcus +\- Bottle Dregs
One method that can be used to establish a minimum level of acidity in a sour beer while also allowing it to enjoy the flavor possibilities afforded by a robust mixed culture is to use a three step fermentation process in which Pediococcus and/or bottle dregs are added along with Brettanomyces in the last step.
Spontaneous Fermentation
Spontaneous fermentation, a term used by brewers to describe the inoculation of wort with an unknown assortment of microbes from the air, is the classic method used to produce lambic beers. As an overview: In spontaneous fermentation a wort is pumped hot into a large, shallow, open air cooling vessel called a koelschip (pronounced and sometimes spelled “coolship”) and allowed to cool overnight to near room temperature. During this time the wort becomes exposed to a variety of microbes floating in the open air. The following day, the wort is pumped into a homogenization tank and then loaded into wooden vessels (oak or chestnut barrels, or larger vessels such as foudres) to undergo both primary fermentation and aging.
Because this method takes strain selection and pitch rates out of our control it is easily the least predictable of the three major options presented here. In classic lambic beers, an early bloom of bacteria that would be considered food spoilers such as Enterobacter and Klebsiella are soon replaced with a vigorous fermentation by Saccharomyces. The modest pH lowering of Saccharomyces and its production of alcohol prevent further activity by bacteria that could potentially cause illness. After Saccharomyces reaches the limits of its ability to attenuate the beer, both Brettanomyces and Pediococcus take over and begin a long and slow period of conditioning during which dextrins are further consumed, lactic acid is produced, and a wide variety of flavors and aromas emerge.
The idea that the flavors of traditional lambic are impossible to achieve through spontaneous fermentation in areas outside of the Senne Valley in Belgium is largely a myth. Additionally, I think that it is a myth that such flavors require spontaneous fermentation to develop at all. So where does spontaneous fermentation (outside of lambic production) fit within a modern sour brewing culture? I think the answer to this is two-fold. The first benefit of spontaneous fermentation is a cultural connection to a more ancient brewing process, one which gave us our first sour beers. The second benefit to this method is the ability to capture and utilize strains of microbes that are not available from a lab, they are truly local and will produce unique beers.
Due to the very nature of spontaneous fermentation, there isn’t much to be done to manage these fermentations aside from the control of environmental factors such as temperature and oxygen exposure. While it is not the purview of this article, the majority of steps used to control spontaneous fermentation are employed on the brew day and through recipe design. We will cover all of these in detail in the upcoming third article of this series.
The use of small test batches to collect wild cultures is one method that I think can help to give us the benefits of wild microbes without all of the major drawbacks (when things go wrong, spontaneous beers can be riddled with off-flavors, unwanted microbes like mold, or can be downright undrinkable). Using this method, small batches of wort are allowed to spontaneously ferment. These batches are then measured for attenuation and pH drop and then tasted. The ones that turn out well can be grown up as mixed cultures to be used for larger batches or employed in staggered additions in the same way bottle dregs can be used.
Methods for Measuring and Managing Sour Beer Fermentations
Now that we have an understanding of the microorganisms involved in sour beer production and a variety of methods used to introduce them into our worts, this final portion of the article will discuss measurements and adjustments that can be made to a fermenting or aging sour beer:
Measuring Gravity
One of the most universal assessments of a fermentation’s progress is the measurement of sugars remaining in the beer. This is generally achieved in one of two ways: by measuring the optical rotation of light passing through a sample or by measuring the density of a sample. The first method is achieved through the use of a refractometer. Refractometers have the benefit of being quick to use and requiring very low volumes of sample wort. Unfortunately, their readings become inaccurate once lactic acid or alcohol are present in the solution, so these tools are only recommended for use during the brew day.
The measurement of wort or beer density is typically achieved via a classic (glass) or digital hydrometer. Glass hydrometers are suspended in a testing cylinder filled with wort or beer, whereas the much more expensive digital hydrometers draw up a small sample for analysis via built-in pipette. The measurement these devices give is accurate regardless of the beer’s stage of fermentation. It is important to remember that all forms of gravity measurement are based on a calibration temperature for your device. If the sample is colder or warmer than your calibration temperature, a conversion factor must be applied to get an accurate measurement.
It is worthwhile to mention that the transformation of sugar into lactic acid does not have a significant effect on the density of a wort. A statistically significant change in the density of a beer only occurs when carbon dioxide is evolved from solution. Therefore, a change in gravity has the same correlation to alcohol production for sour beers as it does for non-sour beers. The only exception to this would be the conversion of ethanol to acetic acid, which does not evolve CO2. Beers with a higher proportion of acetic acid may, in turn, have slightly lower true ABV measurements than the calculations using change in gravity would indicate. In these cases, ABV can be measured in a lab using fractional distillation.
Gravity measurements give us a window into how actively the microbes in a sour beer are metabolizing. The calculation of attenuation allows us to decide whether a fermentation is proceeding and allows us to estimate about how much further it may go. When it comes time to blend or package a beer, consistency in gravity measurements over a period of 6 to 8 weeks tends to be an indication that the beer has stabilized, and the risk of over-carbonation in the bottle is minimized.
Adjusting Gravity
While the gravity of a wort is typically established on brew day, there are a few situations where you may make adjustments to a fermenting or aging beer’s gravity:
- A beer is near 100% attenuation, but lacks fermentation character – While Brett can continue to create flavor changes in a fully attenuated beer, it often does so faster if it has a little sugar to feed its growth. In these cases, you may choose to add a fresh culture of Brett while also adding more sugar to the beer in the form of fresh wort, a sugar source like honey or table sugar, or via an ingredient addition like fruit.
- A beer has a stable final gravity that seems too high – In some cases, the strains of microbes we have chosen may not attenuate the beer enough for the style / our goals. This can occur because certain strains of Lacto, Brett, or Pedio may lack the necessary enzymes to further attenuate the beer. In these cases, it is best to add a fresh culture to the beer, choosing a strain that is known to be able to fully attenuate a dextrinous beer.
- A beer’s ABV is higher or lower than desired – While most craft brewers focus on flavor and not alcohol measurements, in some cases legal requirements or the goals of the brewer may necessitate an adjustment to the final ABV of a beer. These adjustments can be made by adding sugar sources, adding water, or blending with a different ABV beer.
Measuring pH and Titratable Acidity
pH and titratable acidity (TA) are two measurements that describe different aspects of the acidity within a sour beer. The sour flavors that we taste in these beers are the result of a variety of organic acids. The most common of these are lactic and acetic acid, but, depending upon the fermentation and any specialty ingredients, malic, tartaric, citric, and other less common acids may also be present. All acids are designated as such due to their ability to release one or more free protons (H+ Ions) into an aqueous solution. All of the acids in sour beers are considered to be weak acids, meaning that some portion of the total molecules present will have released their proton(s) into solution, while the remainder will have not. This equilibrium concept lies at the heart of the difference between pH and titratable acidity.

I personally use the Milwaukee MW102 pH meter with automatic temperature compensation. I find this to be an accurate, reliable, and easy to use meter.
pH is a measurement of the number of free protons in solution. This number, while constant for any given beer, is decided both by the quantity of acid molecules present AND by the quantity of a wide variety of buffering compounds. Buffering compounds in beer can include mineral ions from brewing water as well as compounds released from malt, hops, the microbes fermenting the beer, and any special ingredients. Therefore, pH is an indirect measurement of how many acidic molecules are in a beer, and as such, this measurement does not correlate exactly to how sour a beer will taste. However, pH is an important measurement because it describes the biologic activity of acids in the beer. Microorganisms don’t care about acid molecules that haven’t released a proton, they only care about the protons themselves. This is due to the fact that the free protons have an effect on a wide variety of chemical reactions, ranging from the activity of enzymes to the ability for a cell to metabolize, reproduce, or survive.
On the other hand, Titratable Acidity (TA), is a measurement that determines the actual quantity of acid molecules in a beer (in any solution). This measurement uses a chemical called a strong base to forcibly remove all of the protons from all of the acid molecules in a sample of beer. By knowing how much strong base we had to add to the beer to achieve this, we can know how much acid was in the beer to begin with. Unlike pH, TA correlates strongly to how much sourness we taste in a beer.
When it comes to sour brewing, an accurate pH reading can only be achieved with an electronic pH meter. Those pH test strips used to measure mash pH (even the really good ones) aren’t accurate enough to tell us anything useful about the pH of our sour beers during fermentation. Like gravity measurements, the pH of our beer is also affected by the temperature of that beer, so choosing a pH meter with a secondary temperature probe and automatic temperature adjustment is beneficial. Otherwise, click here for a calculator to do this correction.
 When measuring TA, we will also be using a pH meter with a few extra steps. To perform this measurement, we will take an accurately measured sample of beer, de-carbonate it, and then insert a pH measurement probe into the sample. At this point, using a calibrated liquid measurement device such as a pipette, we will begin adding a solution of 0.1 N NaOH (sodium hydroxide, our strong base) to the beer sample until the pH reaches 7. We will then slowly continue to add the reagent until the sample reaches 8.2 pH. Make sure to thoroughly stir the sample after each addition of reagent to ensure an accurate reading. Record both volumes of NaOH used and add them together. Once you have recorded the original volume of the sample as well as the total volume of NaOH needed to reach pH 8.2, you can use the calculator included here to determine TA.
When measuring TA, we will also be using a pH meter with a few extra steps. To perform this measurement, we will take an accurately measured sample of beer, de-carbonate it, and then insert a pH measurement probe into the sample. At this point, using a calibrated liquid measurement device such as a pipette, we will begin adding a solution of 0.1 N NaOH (sodium hydroxide, our strong base) to the beer sample until the pH reaches 7. We will then slowly continue to add the reagent until the sample reaches 8.2 pH. Make sure to thoroughly stir the sample after each addition of reagent to ensure an accurate reading. Record both volumes of NaOH used and add them together. Once you have recorded the original volume of the sample as well as the total volume of NaOH needed to reach pH 8.2, you can use the calculator included here to determine TA.
2017 Update: The generalized equation previously provided has been updated to a more accurate and specific calculator. This calculator determines the moles of NaOH required to equalize the pH of the sample and converts that number automatically into the TA measurement for a number of common acids. TA measurements are reported in grams/Liter TA and are specific to the acid being measured. In sour brewing, we assume the majority of acid in the beer is lactic acid and report the titratable acidity as “g/L Lactic Acid”.
For more information on performing a TA measurement, check out the Milk The Funk page on the subject.
It’s important to keep in mind that while TA does correlate strongly to how sour a beer will taste, it is still not the end-all-be-all determiner of sour flavor. Factors such as IBUs, hop flavor, final gravity, melanoidin content, malt flavors, minerality, fermentation flavors, and specialty ingredient flavors all affect how we will perceive the sourness of a beer. Numbers and calculations are useful, but sour beers aren’t created in a computer. Sensory assessment of aroma and flavor by a taster or group of tasters will always be the gold standard for the creation of excellent sour beers.
Titratable Acidity Calculator
Adjusting Acidity
There are a variety of situations where acidity control or adjustments are desired in sour brewing. Examples include:
- A Lactobacillus fermentation has reached a point where the acidity is just right, no further acidification is desired. In these cases, the wort can be re-boiled to kill off the souring bacteria, or a dose of dry hops can be added to the fermenting beer to arrest Lactobacillus metabolism.
- A wort has become too acidic to fully attenuate (typically pH 3.0 or lower). In these cases, it is necessary to blend in fresh wort or another fermented beer in order to raise the blended pH and allow organisms like Brettanomyces to continue fermenting the residual sugars. It is a good idea to build some IBUs into the blending beer as to inhibit further Lactobacillus activity.
- An attenuated beer did not develop the acidity that was desired. In these cases, it is best to add a fresh culture of Pediococcus and potentially some additional sugar sources.
- An attenuated beer has become too acidic. These beers are referred to as “acid beers”. While they are not pleasant to drink on their own, they make an excellent blending tool when adjusting the acidity of a final blend.
Measuring Pitch Rate

This is the microscope that I use for cell counting. Features that make this model particularly useful are the 400x objective, mechanical slide adjustment, and digital camera.
The number of microbial cells pitched into a wort or beer is a huge factor in determining both the health and results of a fermentation. In order to perform cell counts, you will need a microscope and a specialized slide called a hemocytometer or Neubauer Chamber. Cell counting is a laboratory skill that is beyond the scope of this article, but with the right tools it is relatively easy to do for both professional and home brewers alike. My purpose here is to instill the importance of pitch rate in fermentation management.
Here are two excellent resources to learn more about cell counting:
- Eureka Brewing – Counting Yeast Cells
- BSG Craft Brewing – Getting the Most Out of Your Hemocytometer

A hemocytomer is a thick glass slide with laser etched grid lines that has been calibrated to hold a specific volume of sample fluid.
Measuring & Adjusting Temperature
Temperature control during various stages of fermentation can have a very large impact on the ability of the microbes involved to healthily achieve our goals without creating off flavors. Fermentation temperature can be measured using a variety of thermometer options. When beer is actively fermenting, it is best to measure the temperature of the beer itself, as metabolic activity can raise the temperature of fermenting beer above the temperature of the fermentation space. During aging, this degree of temperature monitoring is impractical and unnecessary. During this time, it is valuable to measure and / or control the temperature of the barrel room itself or whatever environment in which your aging sour beers are housed.
As a homebrewer, I make use of small chest freezers, fermwraps, and dual-stage temperature controllers to set and maintain my desired fermentation temperatures. I find these to be highly versatile, and able to maintain temperatures anywhere from freezing to 115° F with ease. Some other excellent options for maintaining temperature include:
- Sous Vide Cookers
- Crockpots with Temperature Monitoring (for Lactobacillus starters)
- The Brewjacket
- Temperature Controlled Conical Fermenters
Managing Oxygen Exposure
Oxygen plays a dual role in sour beer fermentations, both as a vital requirement for healthy Saccharomyces and Brettanomyces growth, as well as a potential source of off-flavors. This duality leaves us wanting to optimize oxygen exposure at differing times in a sour beer’s life cycle. Here are some general guidelines regarding oxygen:
- We want thorough wort oxygenation at the early stage of active Saccharomyces or Brettanomyces fermentation. This is vital to cell growth and appropriate levels of cell growth help to develop the fermentation characteristics we are looking for while avoiding off-flavors.
- We want very low, yet present, exposure (micro-oxygenation) during Brettanomyces aging. Unlike Saccharomyces, Brettanomyces cannot actually ferment sugars into ethanol in a completely oxygen free environment. This curiosity, known as the Negative Pasteur Effect, is the reason why these yeasts thrive in oak barrels, an environment that continually allows them access to low levels of oxygen. This is not to discourage the use of carboys or conical fermenters for Brettanomyces aging. In practical applications, it is nearly impossible to create an aging environment so oxygen-free that Brettanomyces can no longer ferment.
- After a beer has matured, or is ready to be packaged, we want to eliminate all oxygen exposure if possible. At this point, oxygen can increase acetic acid production or create staling of malt or hop compounds.
At the beginning of active fermentation, sour beer wort should be oxygenated using the same practices employed for clean beer fermentation. While many homebrewers aerate their wort via shaking or a sintered air-stone, I prefer to employ a small oxygen tank and a sintered stone while swirling the wort for about 1 minute. For even more repeatable results, an oxygen flow meter can be combined with a sintered stone or in-line oxygenation during wort transfer. This last practice is the standard used by professional brewers. Professional brewers may also measure the oxygenation of wort at the beginning of fermentation with a dissolved oxygen (DO) meter. In my opinion, such a meter would be cost-prohibitive and unnecessary for homebrewers.
Regardless of the choice of vessel, minimization of oxygen exposure should be the goal during a sour beer’s aging. When using carboys, this means that after any sampling the headspace should be purged with CO2. When aging in barrels, sampling is typically done via a small “Vinnie” nail installed into the head of the barrel. Professional brewers tend to be split into two camps in regards to whether or not to top-off a barrel after months of aging (beer volume will be naturally lost to evaporation through barrel walls). I have no personal experience with the issue, but can say that I’ve had excellent beers from different breweries using both methods as well as overly acetic beers from breweries using both methods. I therefore tend to believe that topping-off a barrel or not topping it off has much less of an effect than factors such as barrel wall thickness, strain selection, and sampling methods. Regardless of vessel choice, remember to regularly top-off any airlocks (if used). In my experience, a dried up airlock is surely a sign that the beer within has become too acetic (vinegary).
For homebrewers, Cornelius Kegs make an excellent choice for aging sour beer. These vessels can maintain a very low oxygen environment, offer easy sampling, and can be used to ferment under mild pressure which may help to encourage or subdue fermentation character depending upon the microbes employed.
Preventing Unwanted Microbial Contamination & Mold Growth
When thinking about fermentation and aging, we tend to focus on the changes happening within the beer itself and it can be easy to overlook issues occurring outside of the fermentors:
-
If using airlocks, I like to give these a visual inspection and smell the liquid inside every few months. In some cases, I have found these to develop a vinegary smell due to potential contamination with Acetobacter by fruit flies visiting the airlocks. While I haven’t noticed this infection propagate into the beer itself, it certainly could if left unchecked.
- As I just alluded, fruit fly populations should be controlled in aging spaces and barrel rooms because they are a vector for Acetobacter infection. Various traps can be set up to catch them and any spilled or open beer should be cleaned up thoroughly to discourage them from visiting.
- Another reason to clean up spilled sour beer is the potential for Brettanomyces to sporulate. If a Brett strain has the potential to create ascospores, it is much more likely to do so in a slowly drying puddle of beer than inside of a fermentation vessel. While not of immediate concern to aging sour beer, airborne Brettanomyces spores can potentially make their way into clean beer barrels as well.
- Humidity should be monitored and adjusted in barrel rooms both for the benefit of the beer and the humans who work there. While there does not seem to be an exact industry standard among brewers and winemakers, 60% relative humidity is the highest level I will recommend to help minimize beer loss to evaporation. Humidity higher than this increases the risk of mold and mildew growth, which can be hazardous for human health.
 Safely Assessing Flavor
Safely Assessing Flavor
While all of the measurements and methods discussed here can give us important insight into our sour beer fermentations, they are not a replacement for our own palates. I personally taste my fermenting and aging sour beers at every step of the process, and routinely as they age, with only one exception:
I do not recommend tasting any spontaneously fermented beer, beer being inoculated with unknown mixed cultures, or wort in general until at least one of two conditions have been verified:
- The pH has dropped below 4.5
- A minimum of 2% ABV has been established.
Before these conditions are met, you cannot guarantee that the beer may not be harboring potential food borne illness such as E. coli. Listeria, or Salmonella. Classic clean beer fermentations achieve both of these conditions within the first day or two of active fermentation. On the other hand, sour beers being spontaneously fermented or utilizing low-pitch rate inoculants like bottle dregs, may not achieve these conditions for up to several weeks. Therefore it is prudent to take a pH reading and assess what degree of attenuation has occurred before tasting any sluggishly fermenting sour beer.
Once these basic precautions are employed, I highly recommend tasting your sour beers routinely to assess flavor and aroma development. To learn about the best practices for tastings and for a thorough rundown of off-flavors to watch out for, I recommend that you check out the first article in this series: Blending Sour Beers.
Conclusion
To our ancient ancestors, the act of fermentation must have been a mysterious and somewhat magical process. While today we have a greater understanding of fermentation than ever before, I still think that it holds a bit of magic. On a philosophical level, I personally find it to be wonderfully serendipitous that a variety of microorganisms actually evolved the ability to work together to create these delicious beverages. Each of the microbes involved in a mixed culture fermentation are working to achieve their own goals of growth and reproduction. The fact that in doing so, they not only create a byproduct that is complex and tasty but also safe to drink, is a true wonder of nature.
During its writing, I became aware that this is a very long article… to date the longest that I’ve written. While my goal for any article is never to be lengthy, rather to be thorough and informative, I think that for my article on fermentation to be the longest is a reflection of this topic’s relative importance to the creation of delicious sour beers. Many sour beers begin with deceptively simple recipes, yet through the magic of mixed fermentation, these beers can become some of the most complex beverages in the world. I think that many starting brewers tend to place a misguided level of romanticism on recipe design and the brew day. This is not to say that those factors are not important, only that, at least in my mind, they cannot hold a candle to the value of fermentation. With that said, in a few months I intend to write an article all about the sour beer brew day, so please don’t avoid reading that one!
On that (hopefully) humorous note, I will draw this article to a close. Thank you for taking the time to read it, I hope that it has been both educational and enjoyable. As always, I encourage readers to write below with questions, comments, or suggestions. Additionally, I am always happy to converse with you directly through e-mail, so don’t hesitate to contact me. Happy fermenting!
Cheers!
Matt “Dr. Lambic” Miller
References
Basso, Rafael Felipe, André Ricardo Alcarde, and Cauré Barbosa Portugal. “Could Non-Saccharomyces Yeasts Contribute on Innovative Brewing Fermentations?” Food Research International 86 (2016): 112-20. Web.
“Brettanomyces.” Milk The Funk Wiki. Web. 25 Aug. 2016. <http://www.milkthefunk.com/wiki/Brettanomyces>.
Cui, Yanhua, Tong Hu, Xiaojun Qu, Lanwei Zhang, Zhongqing Ding, and Aijun Dong. “Plasmids from Food Lactic Acid Bacteria: Diversity, Similarity, and New Developments.” IJMS International Journal of Molecular Sciences 16.6 (2015): 13172-3202. Web. 25 Aug. 2016.
“Foodborne Germs and Illnesses.” Centers for Disease Control and Prevention. Centers for Disease Control and Prevention, 21 Dec. 2015. Web. 25 Aug. 2016. <http://www.cdc.gov/foodsafety/foodborne-germs.html>.
Guinard, Jean-Xavier. Lambic. Boulder, CO, USA: Brewers Publications, 1990. Print.
Herskowitz, Ira. “Life Cycle of the Budding Yeast Saccharomyces Cerevisiae.” Microbiological Reviews 52.4 (1988): 536-53. Web. 25 Aug. 2016.
Humbard, Matt. “Off Beat Yeasts #1: Kluyveromyces Lactis.” A PhD in Beer., 21 Feb. 2016. Web. 25 Aug. 2016. <https://phdinbeer.com/2016/02/21/off-beat-yeasts-1-kluyveromyces-lactis/>.
Kalac, Pavel, and Martin Krízek. “A Review of Biogenic Amines and Polyamines in Beer.” Journal of the Institute of Brewing 109.2 (2003): 123-28. Web. 25 Aug. 2016.
“Lactobacillus.” Milk The Funk Wiki. Web. 25 Aug. 2016. <http://www.milkthefunk.com/wiki/Lactobacillus>.
Mello, Jeff. “What Is Wild Yeast, Where Is It, and How Do You Brew With It?” National Homebrewers Conference 2016. Baltimore. 10 June 2016. Lecture.
Neiman, A. M. “Ascospore Formation in the Yeast Saccharomyces Cerevisiae.” Microbiology and Molecular Biology Reviews 69.4 (2005): 565-84. Web.
Papagianni, Maria, and Sofia Anastasiadou. “Pediocins: The Bacteriocins of Pediococci. Sources, Production, Properties and Applications.” Microbial Cell Factories 8.1 (2009): 3. Web. 25 Aug. 2016.
“Pediococcus.” Milk The Funk Wiki. Web. 25 Aug. 2016. <http://www.milkthefunk.com/wiki/Pediococcus>.
Petrova, Penka, Kaloyan Petrov, and Galina Stoyancheva. “Starch-modifying Enzymes of Lactic Acid Bacteria – Structures, Properties, and Applications.” Starch – Stärke Starch/Stärke 65.1-2 (2012): 34-47. Web. 25 Aug. 2016.
Scanes, K. T., B. A. Prior, and S. Hohmann. “Glycerol Production by the Yeast Saccharomyces Cerevisiae and Its Relevance to Wine: A Review.” South African Journal of Enology and Vitriculture 19.1 (1998): 17-24. Web. 25 Aug. 2016.
Somkuti, George A., and Dennis H. Steinberg. “Pediocin Production in Milk by Pediococcus Acidilactici in Co-culture with Streptococcus Thermophilus and Lactobacillus Delbrueckii Subsp. Bulgaricus.” Journal of Industrial Microbiology & Biotechnology J Ind Microbiol Biotechnol 37.1 (2009): 65-69. Web. 25 Aug. 2016.
Steensels, Jan, Luk Daenen, Philippe Malcorps, Guy Derdelinckx, Hubert Verachtert, and Kevin J. Verstrepen. “Brettanomyces Yeasts — From Spoilage Organisms to Valuable Contributors to Industrial Fermentations.” International Journal of Food Microbiology 206 (2015): 24-38. Web.
“Sugar Composition of Wort.” Eureka Brewing., 13 Jan. 2015. Web. 25 Aug. 2016. <https://eurekabrewing.wordpress.com/2015/01/13/sugar-composition-of-wort/>.
“Timeline: Organisms That Have Had Their Genomes Sequenced.” Yourgenome.org. Web. 25 Aug. 2016. <http://www.yourgenome.org/facts/timeline-organisms-that-have-had-their-genomes-sequenced>.
Tonsmeire, Michael. American Sour Beers: Innovative Techniques for Mixed Fermentations. Boulder, CO: Brewers Publications, 2014. Print.
“What Is Yeast?” Wyeast Laboratories : Home Enthusiasts : Brewers : Technical Information : Yeast Fundamentals. Web. 25 Aug. 2016. <https://www.wyeastlab.com/he-yeast-fundamentals.cfm>.
White, Chris, and Jamil Zainasheff. Yeast: The Practical Guide to Beer Fermentation. Boulder, CO: Brewers Publications, 2010. Print.